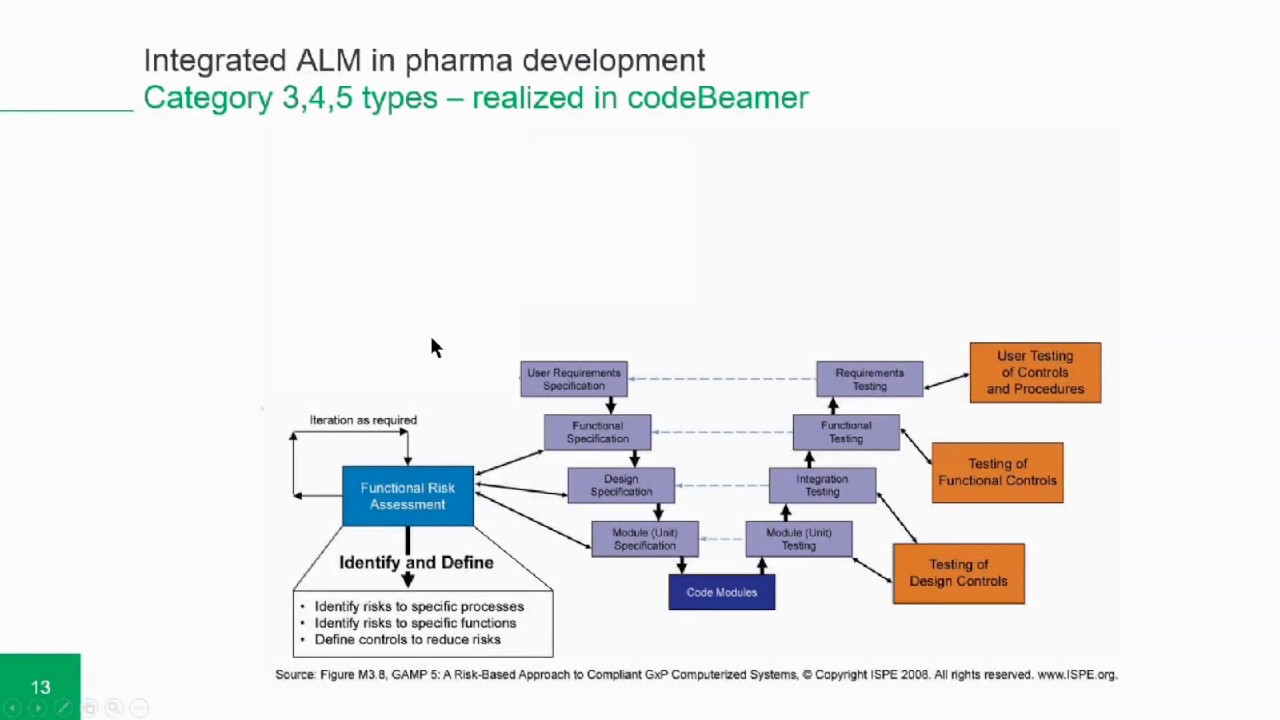 
The GAMP guide has been updated to keep up with concepts and regulatory & industry developments. January 2008 being the latest releases of the GAMP 5 guidelines1- 2. The first GAMP guidelines were put into action in March 1994. GAMP comes in to force For the better of understanding of healthcare automated manufacturing started in the late 80's and early 90's, when greater validation of the pharmaceutical industries was becoming necessary as automated systems played a greater role in healthcare production. Tools should be used accordance with defined instructions, manuals as appropriate, and tool should be held under Configuration Management. Any use of automated test execution tools should defined in test strategy. INTRODUCTION AMP 5, addresses automated testing, states, "Automated test execution tools can used to improve test execution efficiency and effectiveness". Keywords: Computer system validation, EDMS, GAMP, Risk Management. These initiatives can realize significant value by the adoption and integration with the computer system compliance process and EDMS. Should consider a formalized validation plan for each tool or set of tools to describe the risk, use, and validation or qualification Management systems, Laboratory information management systems, Corrective action and Preventive action, etc. GAMP 5 states thatĪutomated computer system validation (CSV) testing tools can be used to improve test execution efficiency and effectiveness.Īutomated CSV tools provide the most benefit for larger enterprise applications such as enterprise resource planning, Document Techniques to allow manufacturers to put their time and effort in electronic data management and testing. As part of its recent initiative cGMPs for the twenty-first century "A Risk-Based Approach",įDA has begun applying risk and impact assessment, including validation of Electronic Data Management System (EDMS),Ĭategorization of system components and supplier assessment as a validation strategy. Revised several times to accommodate changes to regulatory policies, best industrial practice requirements and in computer system Produced by the GAMP Forum, a technical subcommittee of ISPE, the GAMP Guide has been The GAMP Guide is a voluntary set of guidelines created by industry leaders to help companies understand and meet cGMP Vishal Gupta*ĭepartment of Pharmaceutics, JSS College of Pharmacy, JSS University, Sri Shivarathreeshwara Nagara, Mysuru, Karnataka, India. 
Unauthorised republication, reproduction, distribution, 195 Charan H.Y, N. Unauthorised republication, reproduction, distribution, dissemination and copying of this document in whole or in part is strictly prohibited. 34, Pages: 195-198 ISSN 0976 - 044X International Journal of Pharmaceutical Sciences Review and Research Available online at © Copyright protected. Res., 36(1), January - February 2016 Article No.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
